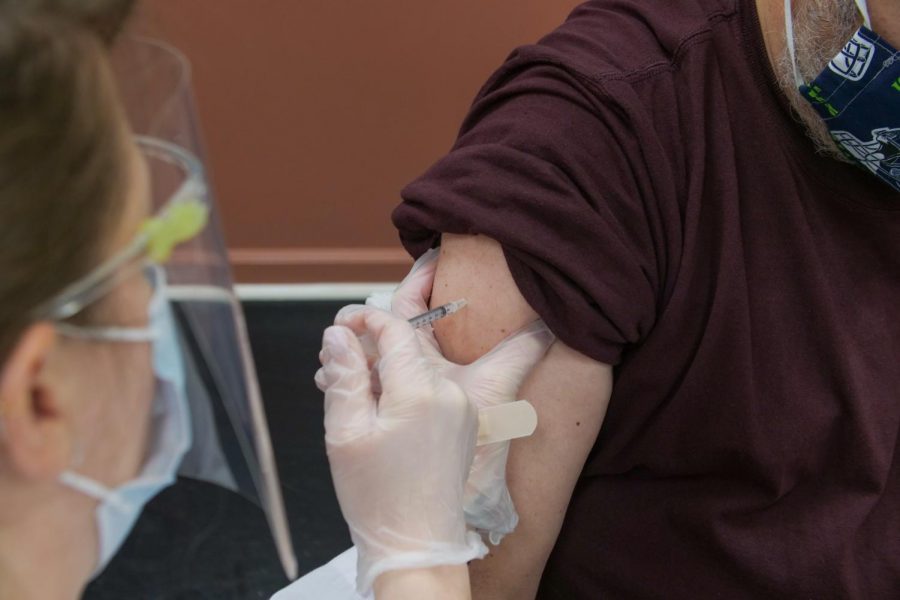
The Pfizer vaccine: a step closer to normalcy
Just like other vaccines, the Pfizer vaccine will be administered to patients in their upper arm. Steven Cornfield/Unsplash
February 17, 2021
The Pfizer-BioNTech COVID-19 vaccine, also known as the Pfizer vaccine, was approved by the Food and Drug Administration in December. Since December, states in America have been distributing the vaccines to residents.
The Pfizer vaccine is an mRNA vaccine that was manufactured by Pfizer, Inc., and BioNTech. This vaccine comes in the form of two shots that must be given in the muscle of a person’s upper arm three weeks apart from each other.
The main and only active ingredient in the Pfizer vaccine is mRNA- messenger ribonucleic acid. mRNA will tell our bodies how to create viral proteins that will trigger our immune response to fight off COVID if we are ever exposed to it.
Note that the Pfizer vaccine will not change the DNA of anybody that receives it.
Common side effects of the Pfizer vaccine include pain, swelling, and redness in the arm area. Patients may also experience headaches, chills, and tiredness. Most of the side effects are not severe or life threatening.
The Pfizer vaccine will be distributed to Connecticut residents in different phases based on age, medical condition, and job.
Phase 1a, the first phase, will have vaccines distributed to those that work in the healthcare field. This includes first responders, healthcare staff, and those that work in long-term care.
In phase 1b, the second phase, vaccines will be given to those that are 75 years and older and those that work in “congregate[d] settings” like homeless shelters and correctional facilities.
Starting on February 11th, those that are between 64 and 75 years old will be scheduled for their vaccine appointment. In March frontline essential workers, like teachers and public transit workers will be able to get their vaccines.
More details on the last phase, phase 1c, will be released soon. During the summer and in the fall of 2021, the vaccine will be available to more of the general public.
Currently those under 16 will not be receiving any COVID-19 vaccines. Children have a different immune system compared to adults. Research on the vaccine has only been done in groups where participants were over 16 years old.
Younger children are typically the last group to be tested on in research trials. Pfzier currently has trials for “children as young as 12.” Vaccines for these groups will be distributed after the FDA has reviewed the data from the newer trials.
“I hope [COVID] will soon fade into memory after the vaccine becomes widely available,” said Tiernan McHugh-Roohr, a Canton High School senior.
After the vaccines are distributed, life will not go back to normal right away. Social distancing and mask wearing will still be required since the vaccine’s effectiveness will build up over time.
For Pfizer’s vaccine, “protection doesn’t start until 12 days after the first shot.” This first shot will only be 52% effective. The Pfizer vaccine will be 95% effective one week after the second shot is given.
Remember: masks, social distancing, and other COVID precautions are not permanent. After enough data is collected from the public, a sense of normalcy will return.